
Download the Galleri Test Requisition Form
If you are a returning provider, sign into the Provider Portal.
Watch a short video of how the test works.
By submitting this form, you agree to GRAIL’s use of this information to contact you, including for marketing purposes. Please do not include any sensitive or confidential information, including health information. For more information, please refer to our privacy notice.
How the Galleri test works
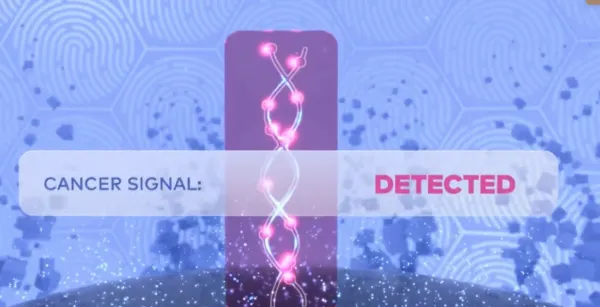
The Galleri test analyzes blood samples for a cancer signal found on cell-free DNA (cfDNA). If detected, Galleri predicts the origin of the cancer signal with high accuracy.1*
Galleri is a screening test and does not diagnose cancer. Diagnostic testing is needed to confirm cancer. Galleri should be used in addition to healthcare provider recommended screening tests
*In the PATHFINDER study, Cancer Signal Origin (CSO) prediction accuracy was 88% for participants with a cancer diagnosis after a "Cancer Signal Detected" test result. Accuracy is the proportion of first or second CSO(s) correctly predicted among true positive participants.
Have a question?
The Galleri test is recommended for use in adults with an elevated risk for cancer, such as those aged 50 or older. The Galleri test does not detect all cancers and should be used in addition to routine cancer screening tests recommended by a healthcare provider. Galleri is intended to detect cancer signals and predict where in the body the cancer signal is located. Use of Galleri is not recommended in individuals who are pregnant, 21 years old or younger, or undergoing active cancer treatment.
Results should be interpreted by a healthcare provider in the context of medical history, clinical signs and symptoms. A test result of No Cancer Signal Detected does not rule out cancer. A test result of Cancer Signal Detected requires confirmatory diagnostic evaluation by medically established procedures (e.g. imaging) to confirm cancer.
If cancer is not confirmed with further testing, it could mean that cancer is not present or testing was insufficient to detect cancer, including due to the cancer being located in a different part of the body. False-positive (a cancer signal detected when cancer is not present) and false-negative (a cancer signal not detected when cancer is present) test results do occur. Rx only.
The GRAIL clinical laboratory is certified under the Clinical Laboratory Improvement Amendments of 1988 (CLIA) and accredited by the College of American Pathologists. The Galleri test was developed and its performance characteristics were determined by GRAIL. The Galleri test has not been cleared or approved by the Food and Drug Administration. The GRAIL clinical laboratory is regulated under CLIA to perform high-complexity testing. The Galleri test is intended for clinical purposes.
- Schrag D, McDonnall CH, Naduld L, et al. PATHFINDER: A Prospective Study of a Multi-Cancer Early Detection Blood Test. Presentation at European Society of Medical Oncology (ESMO) Congress September 9-13, 2022; Paris, France. https://grail.com/wp-content/uploads/2022/09/Schrag_903O_ESMO-2022_Pathfinder-Main_Proferred-Paper-Oral-Presentation.pdf.